Medical Device Vigilance
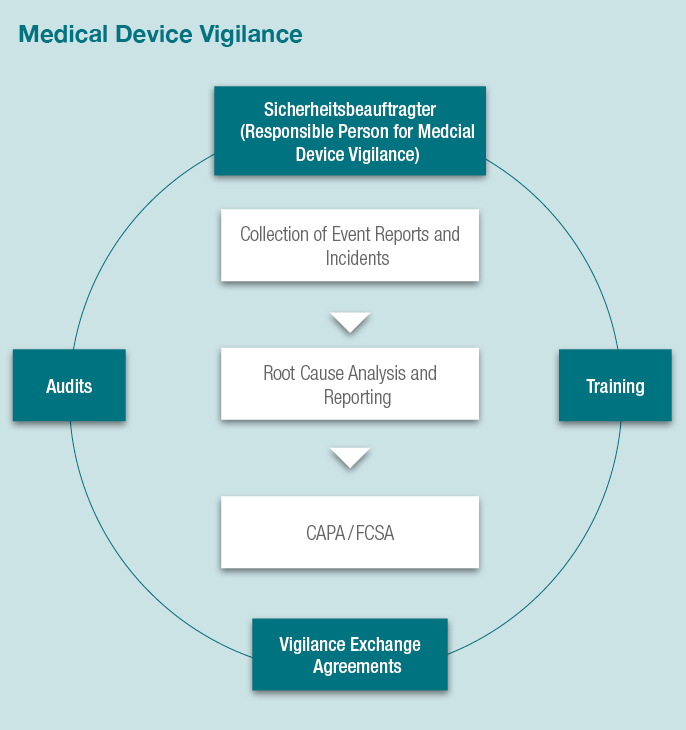
Manufacturers of medical devices are required to monitor the safety of their products by an adequate post marketing surveillance system. We offer you our services for implementation of SOPs and as a Responsible Person for Medical Device Safety (Sicherheitsbeauftragter according to the German MPG).
Our Services
- SOPs are covering the following issues:
- Collection and assessment of events and incidents
- Event reporting
- Trend reporting
- Field Safety Corrective Actions (FSCAs)
- Vigilance Exchange Agreements
- Internal audits and corrective and preventive actions (CAPA)
- Training
- Acting as Responsible Person for Medical Device Safety (Sicherheitsbeauftragter according to the German MPG)
- Clinical Evaluation of Medical Devices
Please do not hesitate to contact us for an individual offer.