Medical and Regulatory Affairs
Environmental Risk Assessment (ERA)
The revised EMA ERA Guideline (EMEA/CHMP/SWP/4447/00 Rev. 1 – Corr.), effective September 2024, modernises the assessment process for the possible environmental impact of human medicinal products. While retaining the established two-tier system, the updated guideline introduces clearer requirements, expands data obligations, and enhances the scientific rigor of assessments. Moreover, EMA, BfArM and other national competent authorities stress the need to update or newly submit an environmental risk assessment (ERA) in the context of new market authorisation applications or variations that affect the environmental exposure.
Phase I now includes an expanded initial screening for all active substances, which determines whether a more in-depth Phase II assessment is required. The guideline also mandates an explicit hazard assessment, with particular attention to PBT (persistent, bioaccumulative, toxic) and vPvB- (very persistent very bioaccumulative) properties.
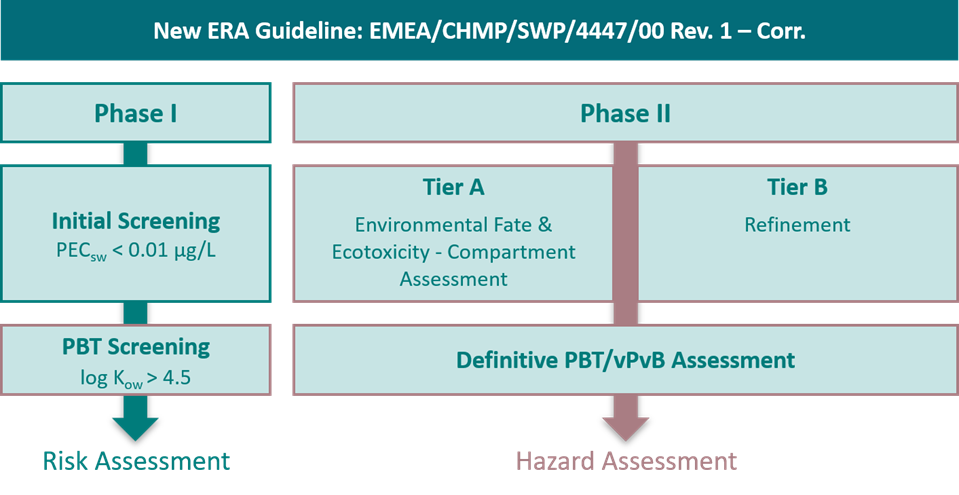
Environmental risk assessment (ERA) according to the new guideline. Phase I starts with an initial screening to assess the predicted environmental concentration in surface water (PECsw). In parallel, the octanol/water partition coefficient (log Kow) is determined as part of the hazard assessment in phase I. Both parameters may trigger the need for Phase II. Phase II is divided into Tier A and B. Tier A comprises the assessment of environmental fate and ecotoxicity, leading to a compartment-specific risk assessment. Tier B includes refinement (higher-tier) studies, if triggered. Overall, phase II supports a definitive PBT/vPvB (PBT: Persistent, Bioaccumulative and Toxic, vPvB: very Persistent and very Bioaccumulative) assessment and a detailed environmental hazard characterisation of the substance.
Implementing the new ERA requirements poses particular challenges for many companies, especially regarding data availability, study design, and interpretation of triggers. We guide you through these requirements efficiently, in regulatory-compliant, cost-effective and proportionate manner.
Dr. Ebeling & Assoc. GmbH provides support not only in the preparation of ERA dossiers but also in the strategic interpretation of the guideline, the identification of robust lines of argument, and communication with regulatory authorities. We advise you throughout all ERA phases – from initial data collection and laboratory studies to the preparation of comprehensive ERA dossiers – while offering strategic advice and continuous monitoring of regulatory developments. For laboratory studies, we collaborate closely with certified, experienced laboratories.
Our ERA services
- Advisory services and portfolio screening
- Gap analyses of existing ERA-relevant data, including structured scientific literature searches and regulatory evaluation of data quality according to CRED and OECD criteria
- Planning, coordination, and regulatory alignment of laboratory studies in collaboration with accredited laboratories following OECD Test Guidelines
- ERA as Add-On to signal management (CoBRA)
ERA Pooling Project in cooperation with the BPI:
In collaboration with the German Pharmaceutical Industry Association (BPI), we offer a synergistic, cost-efficient, and comprehensive solution for the preparation of ERAs that can be used by all MAHs.
Learn more here or contact us if you are interested or require a tailored offer at ERA@ebeling-assoc.com.